Sivaraman Arumugam*,Sadananda Rao Manjunath**,Rajappa Senthilkumar**, Venkaba Rao Srinivasan**,Swaminathan Rajendiran***,Hiroshi Yoshioka****,Yuichi Mori****,Samuel Abraham*****
*Dept. of Arthroscopy & Sports Medicine, Sri Ramachandra University, Chennai, India
**Nichi-In Centre for Regenerative Medicine, Chennai, India
***Central Laboratory, Sri Ramachandra University, Chennai, India
****Waseda University, Tokyo, Japan
*****Yamanashi University- School of Medicine, Chuo, Japan |
Address for Correspondence
Dr. Samuel Abraham
Nichi-In Centre for Regenerative Medicine
C 16&17, Vijaya Health Centre Premises,
175, NSK Salai, Vadapalani,
Chennai -600 026,
Tamil Nadu,
India.
Phone: +91-44-42321322 / 24816743.
Fax No: +91-44- 24732186
Email id: drabrahamsj@ybb.ne.jp
Alternate email id: drspp@nichimail.jp
Abstract
Introduction
Damages in articular cartilage caused by aging, injuries and other factors could lead to osteoarthritis. Success rates of the current conventional strategies for articular cartilage repair are not that encouraging. Autologous chondrocyte transplantation has revolutionized treatment options in cartilage defects in the last decade. Central to their success is the availability of an ideal scaffold. Primary focus of the present study is to develop a suitable non-biological, toxic free and bio-degradable scaffold for autologous cartilage implantation. Earlier studies have concluded that Thermoreversible Gelation Polymer (TGP) is an ideal scaffold for bovine chondrocyte expansion in vitro. Based on this study, we have tested the potential of TGP in supporting in vitro expansion and longer viability of the human chondrocytes in culture without the use of growth factors.
Methods
Human articular cartilage chondrocytes were isolated and embedded in a cylindrical mould of TGP at 8°C and cultured at 37˚C in 5% carbon dioxide incubator up to 12-16 weeks without growth factors. The morphology and characterization of the tissue–scaffold construct was studied by histological Hematoxylin and Eosin staining (H & E) and Immunohistochemical (S-100) staining
Results
The results of H&E and Immunohistochemistry showed positive for chondrocyte markers in the cells grown in TGP.
Conclusion
Based on the results we conclude that the TGP significantly supports the in vitro expansion of the human chondrocytes for a longer period and thus may be considered as a suitable scaffold material for autologous chondrocyte implantation in the future.
J.Orthopaedics 2011;8(3)e5
Keywords:
TGP scaffold; Tissue engineering; Chondrocyte; Articular cartilage; in vitro expansion.
Introduction:
Articular cartilage is highly complex organized avascular tissue in which chondrocytes are embedded in an extracellular matrix. Chondrocytes are responsible for development and maintenance of the cartilage structure (1). The cartilage extracellular matrix (ECM) interacts with chondrocytes through a variety of receptors to modulate chondrocyte metabolism and phenotype (2).
Damages to articular cartilage can be caused by physiological stress, aging, injuries and other factors which lead to osteoarthritis (OA) (3)that eventually requires surgical management at later stages of cartilage destruction(4). Current surgical treatments include tissue response techniques, osteochondral transplantation and cell-based tissue engineering strategies. .Articular cartilages lose self-repair capacity since chondrocytes lose their mitotic ability as early as the first year of life (5). Currently, transplantation of allogeneic cartilage has been reported in many studies but the post implantation viability is minimal (6, 7) Autologous Chondrocyte Transplantation (ACT) is being followed in patients with symptomatic articular cartilage defects with the purpose of replacing the damaged cartilage (8). In this procedure chondrocytes are obtained by arthroscopy from the cartilages from the non-load bearing sites of the patient and cells are cultured for a period of four to six weeks after which the cultured cells are transplanted to the same patient in the area of the defect. This modality has been in practice for more than a decade with numerous clinical trials to support its evidence. A major drawback observed was the formation of fibro cartilage made up of Collagen Type I instead of hyaline cartilage which is the native cartilage tissue of knee joints and the success of the procedure is limited due to relatively shorter viability of tissues formed in vivo.
In this regard recent research has focused on the synthesis of stable cartilage tissues with longer viability to replace the damaged cartilage native to the tissue being replaced. Scaffolds are being employed as cell carriers so that the cells can be positioned at right place, there by ensuring that the cells become evenly distributed in the defect and regenerate the cartilage. Numerous scaffolds including natural scaffolds such as collagen, chitosan, fibrin and synthetic scaffolds like composite, polymers have been employed. Natural scaffolds have the risk of animal protein and prions contamination. This can be overcome by the use of synthetic scaffolds.
Based on the animal model experiments and different supportive scaffolds used previously, we have studied the suitability of Thermoreversible Gelation Polymer (TGP) scaffold for the ex vivo expansion of the chondrocytes. TGP is a non-toxic, bio-degradable scaffold as proved in animal model experiments. This TGP scaffold mimics a native cartilage matrix microenvironment that would contribute greatly to cartilage regeneration and the newly formed tissue engineered cartilage will have characteristics that can be a remarkable alternative for the original cartilage tissue (7).
Yasuda et al in 2006 reported that TGP acts as a 3-D hydrogel scaffold for bovine chondrocytes with longer viability (9). The authors compared the culture of chondrocytes isolated from bovine shoulders in TGP with and without the use of growth factors and the study revealed that though cultures grown with growth factors expressed more Glycosaminoglycans (GAG) and other ECM components when compared to cultures without growth factors in 2 to 3 weeks, however at the end of 12 weeks, there were no differences in the GAG expression between the groups. As a continuum to this study we wanted to study the efficacy of TGP in supporting culture of human articular chondrocytes for a longer period of more than 12 weeks without the use of growth factors.

F(1)
F(2)
F(3)
F(4)
Material and Methods :
Samples
The ethics committee of Sri Ramachandra University, Chennai approved this study. The Cartilage specimens were collected from the articular defect of 8 patients with an average age of 25 years, upper and lower limit of the age was 34 years and 19 years. Based on the guidance and recommendations of the institution’s ethical committee, informed consent was obtained from all the patients. Human cartilage biopsies were obtained by arthroscopy from the non-weight bearing knee cartilage.
Human articular chondrocytes were isolated from the cartilage tissues. The tissue samples were collected in sterile phosphate-buffered saline (PBS) with antibiotics and transported in cool condition to the laboratory for processing.
Chondrocyte isolation
The harvested cartilage was weighed to know the wet weight. The tissues were rinsed with sterile phosphate-buffered saline (PBS) and subjected to digestion with collagenase type II. The minced tissues were mixed with Dulbecco's modified Eagle's medium (DMEM) /Ham’s F12 medium containing 0.3% Collagenase Type II without fetal bovine serum (FBS) and incubated in 5% carbon dioxide (CO2) incubator at 37˚C with an orbital shaker for 12 to 18 Hrs. The digested suspension was filtered with a 70µm nylon mesh; undigested tissues were discarded and filtrate was centrifuged at 1000 rpm for 10 minutes. The cell pellets were washed with PBS and the cell population was counted by trypan blue dye exclusion method (9).
TGP preparation/reconstitution
TGP is a copolymer composed of the Thermoreversible Polymer (Poly (N-isopropylacrylamide-co-n-butylmethacrylate) (polyNIPAAM-c0-BMA)) and the hydrophilic polymer (poly ethylene glycol (PEG)) (9). We procured the TGP in lyophilized form from Nichi-In Biosciences, Chennai, India. This was dissolved in 10 ml of DMEM-Ham’s F12 medium and kept at 4˚C overnight (9).
Chondrocyte culture
Isolated chondrocytes were counted and cultured in two different groups. 1. Cells cultured in the TGP scaffold (group 1) and 2. Cells Cultured without TGP Scaffold (group 2). In each group, 1 million cells per ml were seeded. 1 ml of TGP was mixed with cell pellet and the TGP-chondrocytes cell suspensions were kept below 8˚C to maintain the liquid state of the copolymers and placed into a syringe for injection into a cylindrical silastic tube which was used as a mould. The tube and its contents were warmed above 8˚C in order to solidify the biopolymer, yielding a cylindrical structure (Figure (fig). 1). The chondrocyte-TGP hydrogel cylinders were removed from the silastic tubes and cultured in DMEM-HAM-F12 medium supplemented with 10% FBS, 1% antibiotic-antimycotics and 25µg/ml L-ascorbic acid at 37°C for up to 16 weeks. The culture dishes were placed on the orbital shaker, providing gentle agitation (9). The medium was changed twice a week. Initially the specimens from each group were harvested at 4, 8, 12 and 16 weeks for the following analyses .Later this was optimized for 12 and 16 weeks.
Chondrocytes harvest
After 4, 8, 12 and 16 weeks, the cells were harvested from TGP. The scaffold was cooled below 8˚C kept inside the refrigerator. By this procedure the TGP liquefies and the material remaining will be the cells and or tissues. The obtained harvest was rinsed with PBS and then analyzed for histological staining (10).
Histological examinations
The harvested specimens were fixed in 10% formalin embedded in paraffin and sectioned. With standard histological techniques, serial sections were deparaffinized and stained with hematoxylin and eosin (H&E).Immunohistochemistry studies was done to verify the expression of S-100 protein (a chondrocyte-specific marker) by the cultured cells (11). Cells were attached onto the chamber slides, fixed with 4% paraformaldehyde and washed with Tris-buffered saline twice. The cells were blocked for 10 min in 3% H2O2, then 20 min in 20% FCS in Tris-buffered saline, before incubation with primary antibodies: a mouse anti-S-100 (1:100 for 1h at 37˚C). The antibodies were visualized by a streptavidin-biotin-complex technique according to Zheng et al 2004 (12) protocol. As a negative control for immunohistochemical staining, the primary antibody was replaced by normal mouse or rabbit serum or Tris-buffered saline.
Results
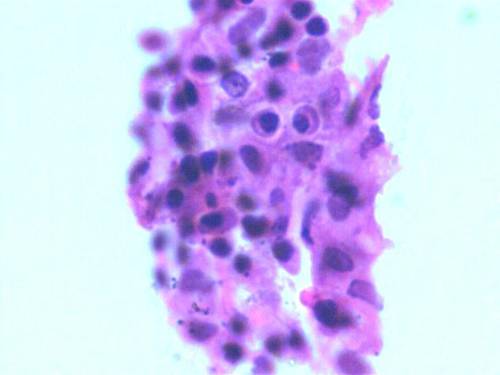
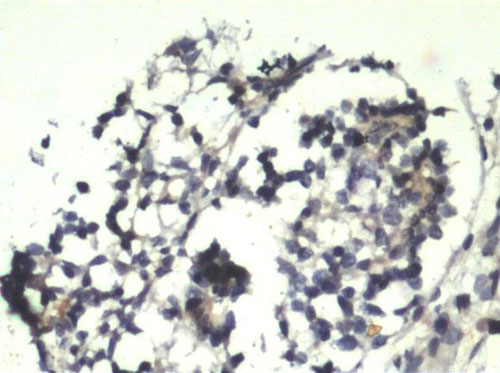
Human chondrocytes were isolated from a total number of 8 samples (n=8). Among the isolated specimens, 6 samples were successfully cultured in in vitro condition. After their in vitro expansion, chondrocytes were harvested and the cell population count and viability was checked by Trypan blue dye exclusion method. Fig. 2 shows the monolayer cells grown after 4 weeks. We found that in the entire successive expanded specimens, cell counts were increased compared to the initially seeded quantity but at the end of the 4th week, the cells seeded without TGP (group 2) lost their viability. Group1 (TGP-chondrocytes), showed good cell number and longer viability which maintained until the end of 16 weeks. In both groups culture conditions were optimized without using growth factors. The harvested cells from group I was positive for the Hematoxylin and Eosin staining (fig. 3). Similarly cells were detected using Immunohistochemistry chondrocytes specific protein marker S-100 (fig. 4).
Discussion :
Chondrocytes or cells of the cartilage are predominantly derived from mesenchymal precursor cells. Tight regulation of chondrocyte differentiation is essential for normal skeletal development, growth, and for the maintenance of joint health (11).
Cartilage damage results in osteoarthritis. Autologous chondrocyte transplantation is a widely used method for cartilage replacement but the stability and the post-implantation viability of the cartilage tissue is limited. Hence use of scaffolds for synthesis of stable cartilage tissues is gaining momentum.
In this regard the aim of this study was to confirm whether TGP is a suitable scaffold for culture of human chondrocytes as Yasuda et al (9) have already confirmed TGP to be an ideal scaffold for bovine chondrocytes in their study.
The characterization results of the present study suggests that the TGP is a suitable material for the ex vivo and in vitro expansion of the human chondrocytes. As like other scaffold models used in different animal model experiments and clinical trials (13-16), TGP scaffold supports the in vitro expansion and it has some of the unique properties like allowing the cells to grow in three dimensional fashion and this scaffold provides the longer viability to the expanded cells without any growth factors (10). Currently in majority of the cases, animal proteins are being used as growth factors, which might have the potential of biological contamination when used in the human clinical trials. So the development of cost effective and non-toxic, degradable synthetic materials like TGP scaffold is essential. Synthetic materials are free from prions/animal protein contamination thereby ensuring safe clinically usable tissues and materials.
The challenge for cartilage tissue engineering is to produce cartilage tissue with suitable structure and properties ex vivo, which can be implanted into joints to provide natural repair that with time, will become integrated with the patient’s tissues. In majority of the studies reported earlier in vitro or ex vivo expansion of the chondrocytes were induced by exposure to selected growth factors. In this study the cultured chondrocytes in TGP, a 3D scaffold have shown longer viability when compared to conventional cell cultures even without the use of growth factors. TGP allows the apparent mobilization of chondrocytes inside the 3D scaffold and this engineered cartilage may help to heal the damages in osteoarthritic joints. In conventional methods, chondrocytes may dedifferentiate into fibroblast-like cells whereas the TGP mediated chondrocyte expansion does not allow this dedifferentiation.
Conclusion
From these observations we conclude that the non-biological, degradable, non toxic, novel TGP scaffold has supported the in vitro/ ex vivo expansion of the human chondrocytes without the use of growth factors. This strategy can be explored further to create longer viable cartilage in vitro and successfully transplant them in vivo for optimal cartilage repair in osteoarthritis and other cartilage defects.
Acknowledgements
The authors acknowledge
- Dr. Deedepiya Devaprasad and Dr. S .P. Preethy for their valuable assistance in preparing this manuscript.
- M/S Hope Foundation (Trust), Chennai, India and M/S Arthroscopy Study Group Foundation (Trust), Chennai, India for funding the study.
Reference :
- Montrull HL, Brizuela NY, Demurtas SL, Spitale L, Meirovich CI: Structure and secretory activity of cultured chondrocytes from patients with osteoarthritis. Biocell 2005, 29 (2):163-7.
- Genes NG, Rowley JA, Mooney DJ, Bonassar LJ: Effect of substrate mechanics on chondrocyte adhesion to modified alginate surfaces. Arch Biochem Biophys 2004, 422:161-7.
- Alexopoulos LG, Erickson GR, Guilak F: A method for quantifying cell size from differential interference contrast images: validation and application to osmotically stressed chondrocytes. J Microsc 2002, 205:125-35.
- Boileau C, Amiable N, Martel-Pelletier J, Fahmi H, Duval N, Pelletier JP: Activation of proteinase-activated receptor 2 in human osteoarthritic cartilage upregulates catabolic and proinflammatory pathways capable of inducing cartilage degradation: a basic science study. Arthritis Res Ther 2007, 96: R121.
- Robinson D, Halperin N, Nevo Z. Fate of allogeneic embryonal chick chondrocytes implanted orthotopically, as determined by the host's age. Mech Ageing Dev. 1989 Oct;50(1):71-80
- Gao J, Yao JQ, Caplan AI: Stem cells for tissue engineering of articular cartilage. Proc Inst Mech Eng [H] 2007, 221: 441-50.
- Robinson D, Ash H, Aviezer D, Agar G, Halperin N, Nevo Z: Autologous chondrocyte transplantation for reconstruction of isolated joint defects: the Assaf Harofeh experience. Isr Med Assoc J 2000, 2 :290-5.
- Vukivevic S, Jelic M, Haspl M: Cultivation and implantation of chondrocytes. Acta Med Croatica 2007, 61(5):453-7.
- Yasuda A, Kojima K, Tinsley KW, Yoshioka H, Mori Y, Vacanti CA. In vitro culture of chondrocytes in a novel thermoreversible gelation polymer scaffold containing growth factors. Tissue Eng 2006; 12:1237-45.
- Kisiday J, Jin M, Kurz B, Hung H, Semino C, Zhang S, Grodzinsky AJ: Self-assembling peptide hydrogel fosters chondrocyte extracellular matrix production and cell division. implications for cartilage tissue repair. Proc Natl Acad Sci U S A 2002; 99:9996-10001.
- Nakamura S, Nakamura T, Kawahara H. S-100 protein in human articular cartilage. Acta Orthop Scand 1988; 59: 438-40.
- Zheng MH, King E, Kirilak Y, Huang L, Papadimitriou JM, Wood DJ, Xu J. Molecular characterisation of chondrocytes in autologous chondrocyte implantation. Int J Mol Med 2004; 13:623-8.
- Henson FM, Vincent T. Chondrocyte outgrowth into a gelatin scaffold in a single impact load model of damage/repair - effect of BMP-2. BMC Musculoskelet Disord 2007; 5; 8:120.
- Wang KH, Kao AP, Wangchen H, Wang FY, Chang CH, Chang CC, Lin SD. Optimizing proliferation and characterization of multipotent stem cells from porcine adipose tissue. Biotechnol Appl Biochem 2008; 51 (Pt 4):159-66.
- Lin YJ, Yen CN, Hu YC, Wu YC, Liao CJ, Chu IM. Chondrocytes culture in three-dimensional porous alginate scaffolds enhanced cell proliferation, matrix synthesis and gene expression. J Biomed Mater Res A 2009; 88(1):23-33.
- Gong Y, Ma Z, Zhou Q, Li J, Gao C, Shen J. Poly(lactic acid) scaffold fabricated by gelatin particle leaching has good biocompatibility for chondrogenesis. J Biomater Sci Polym Ed 2008; 19(2):207-21.
|






