|
Tomoki
Nakamura*, Katsuyuki Kusuzaki*, Takao Matsubara*, Haruhiko
Satonaka, Ken Shintani*, Touru Wakabayashi*,Akihiko
Matsumine*, Atsumasa Uchida*
*Department of
Orthopaedic Surgery, Mie University Faculty of Medicine, Tsu, Mie,
Japan
Address for Correspondence:
Katsuyuki
Kusuzaki MD, Department of Orthopaedic Surgery,
Mie University Faculty of Medicine,
Edobashi 2-174 Tsu Mie 514-8507, Japan
e-mail: kusu@clin.medic.mie-u.ac.jp
Tel: +18 59 231 5022
Fax: +81 59 231 5211
|
|
Abstract:
We
recently encountered a 6-year-old boy with Alagille syndrome,
which is characterized by remarkable hyperbilirubinemia caused
by cholestasis due to the paucity of interlobular bile ducts in
the liver. This patient had a pathological fracture of the femur
with local bone atrophy, with malunion and insufficient callus
formation. Thus, after liver transplantation, we performed a
correction osteotomy. Macroscopically, the femur was stained
dark green and histology of the resected bone at the site of the
malunion revealed the presence of many histiocytes and
osteoclast-like multinucleate giant cells containing bilirubin
particles in the cytoplasm. The multinucleate giant cells were
found to have caused bone resorption. These findings suggested
that bilirubin might activate macrophages to form osteoclast-like
multinucleate giant cells, resulting in bone resorption and
osteoprosis.
J.Orthopaedics 2007;4(2)e17
Introduction:
Recently,
we encountered a rare case of Alagille syndrome, which manifests
as severe jaundice due to cholestasis caused by a paucity of
interlobular bile ducts in the liver associated with skeletal
malformations and cardiovascular dysfunction. The patient, who
was a 6-year-old boy, had been known to have marked
hyperbilirubinemia since birth. He was found to have a
pathological fracture of the shaft of the femur, with malunion
due to local bone fragility.
In this context, it has been reported previously
that hyperbilirubinemia inhibits osteoblast proliferation
and induces osteoprosis6).However, the findings in
this case suggested the possibility that hyperbilirubinemia may
also activate histiocytes or induce the formation of osteoclast-like
multinucleate giant cells. There are no reports in the
literature of the histological findings in the bone in patients
with marked and persistent hyperbilirubinemia.
Case
Report:
The patient was a 6-year-old Indonesian boy. He was
taken to the city hospital in Bali at the age of 9 months for
persistent jaundice. He had the characteristic face of Alagille
syndrome and the liver biopsy findings confirmed the case as one
of Alagille syndrome. Although the patient was then initiated on
vitamin ADEK supplementation, ursodeoxycholic acid and
rifampicin, the liver dysfunction and hyperbilirubinemia had
worsened by the time the patient was 5 years old.
He was referred to our hospital for living- donor
liver transplantation from his father, after getting private and
official financial support from both Japan and Indonesia. Two
months prior to his visit to our hospital, he had developed a
pathological fracture of the left femoral shaft caused by mild
trauma (slipping) and had been given a cast for treatment.
However, he could not walk due to the development of an antero-lateral
convex deformity caused by delayed union. At the initial visit
to our hospital, he was found to be malnourished and exhibited
stunted growth, with a height of 80 cm and body weight of 12 kg.
He had all of the diagnostic features of Alagille syndrome,
including the characteristic face, mild peripheral pulmonary
artery stenosis, butterfly vertebrae, posterior embryotoxon, and
hyperbilirubinemia. Blood examination revealed anemia
(hemoglobin, 9.2g/dl) and liver dysfunction with high serum AST
(223U) and ALT (151U), hyperbilirubinemia (12.8mg/dl), high
serum total cholesterol (909mg/dl), and high serum ALP (2736U).
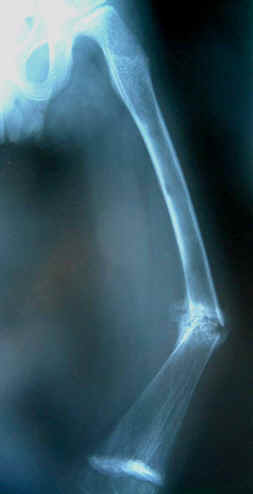
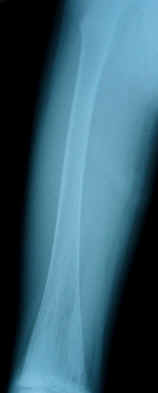
A plain x-ray of the left thigh revealed an antero-lateral
convex deformity of the left femoral shaft caused by malunion of
a fracture with poor callus formation, cortical bone atrophy and
osteoprosis, associated with osteolytic lesions (Fig. 1).
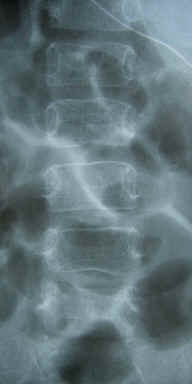
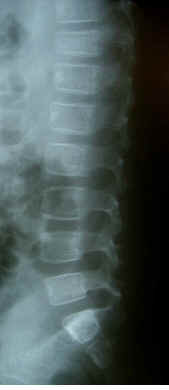
Osteoprosis of the right femur and spine was also found (Fig.
2). The patient could not move without a wheel- chair. After
successful liver transplantation, his parents were anxious for
the fracture also to be treated so that the patient could start
walking before they returned to Indonesia. Because of
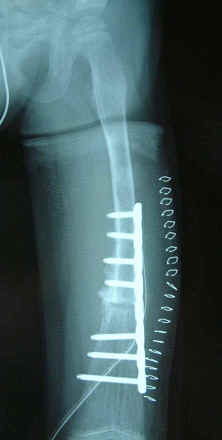
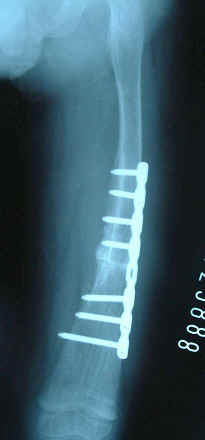
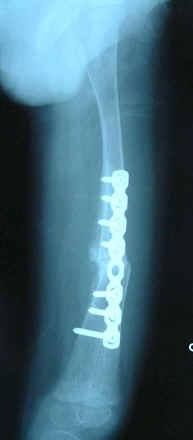
satisfactory postoperative recovery of his liver condition, we
decided to perform a correction osteotomy and osteosynthesis
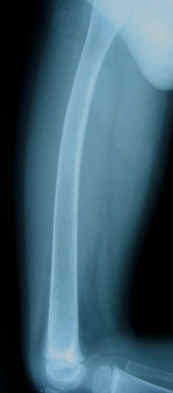
with internal fixation using a plate and screw. After 6 weeks,
partial weight bearing became possible because of complete bone
union (Fig. 3), and finally, by two months later, the patient
could walk without any support.
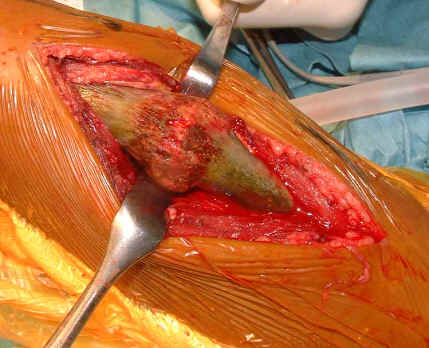
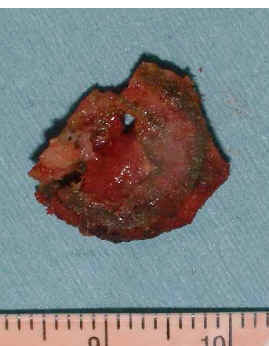
During the osteotomy, the femoral cortex and callus
around the fracture site were macroscopically observed to show
dark green staining a very unusual finding, suggesting bilirubin
deposition. Coronal sections of the femoral shaft also showed
deep green staining of the cortex as well as the callus (Fig.
4).
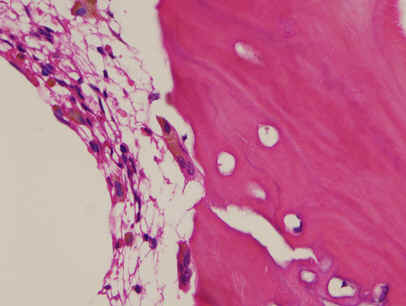
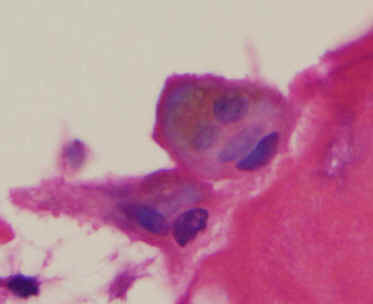
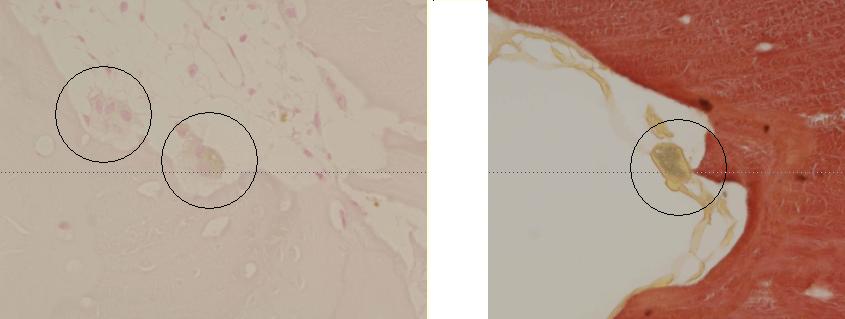
Histopathologically, numerous osteoclast-like
multinucleate giant cells were observed at the surface of the
lamellar bone of the cortex and callus, associated with marked
histiocytic infiltration of the bone marrow. These cells
contained brown pigment in the cytoplasm (Fig. 5). The pigment
was identified as bilirubin based on the negative staining with
Berlin-blue and positive staining by Halls method, which is
specific for bilirubin (Fig. 6).
|
|
|
|
Figure
4: Macroscopic finding of the femoral bone at fracture
site (A) and cross section of resected bone (B) during
surgery of correction osteotomy.
The femoral cortex and callus around the fracture
site macroscopically shows dark green color. The coronal
section of the femur also shows deeply dark green color in
cortex as well as callus
|
|
|
|
|
Figure 5: Histological findings
of resected bone (H.E. A, B: x10 objective lens C: x40
objective lens) There
were many multinuclear giant cells like osteoclast at
surface of lamellar bone of the cortex and callus and many
histiocytes in the bone marrow. These cells contained
brown pigments in the cytoplasms (arrows) |
|
|
|
Figure
6: Histochemical findings of intracellular pigments
after Berlin-blue staining (A) for hemosiderin and
Halls method for bilirubin(B) The intracellular
pigments were not stained with Berlin-blue but stained by
Halls method. |
Discussion :
Alagille syndrome is a very rare disease, occurring
in 1 in 100,000 births, with an equal gender incidence. An
autosomal dominant pattern of inheritance with low penetrance
and a great variability of expression have been reported. The
Alagille gene has been identified in the 20pl2 region. The
syndrome manifests as a multi-system disorder involving the
liver, heart, eyes, face, and skeleton. Since it is
characterized by cholestasis because of a paucity of
interlobular bile ducts 1), 2),
patients frequently have hyperbilirubinemia.
It is well known that patients with chronic liver
disease have an increased prevalence of osteoporosis because of
calcium malabsorption caused by low levels of 25-hydroxy vitamin
D3 and hyperbilirubinemia3). However, the underlying
mechanism of the relation of osteoprosis to hyperbilirubinemia
is still unclear. Guanabens et al reported that patients of
primary biliary cirrhosis with osteoporosis had higher serum
bilirubin levels than those without osteoporosis4).
Ormarsodottir et al also reported that a high serum bilirubin
level was associated independently with increased bone loss at
the femoral neck in patients with chronic liver disease5).
Janes et al demonstrated experimentally that exposure to
excessive levels of bilirubin inhibited the proliferation of
osteoblasts in cell culture6). However, there are no
reports on the relation between the bilirubin level and
osteoclast function.
The present case with long-term hyperbilirubinemia
showed radiographic evidence of systemic osteoprosis and had a
pathological fracture of the femur due to local bone fragility.
During surgical correction of the malunited fracture in the
femur by osteotomy, we found the femur to be stained dark green,
a very unusual finding indeed. This macroscopic color was very
similar to that of condensed bile juice. Histological
examination revealed the presence of numerous histiocytes in the
bone marrow and osteoclast-like multinucleate giant cells lining
the resorbed bone surface in the femoral cortex. Both cells
contained large amounts of brown pigment in the cytoplasm.
Histochemical study revealed that these particles were bilirubin
and not hemosiderin or other pigments derived from iron. The
multinucleate giant cells closely resembled osteoclasts,
however, the number of nuclei was smaller and the cell size was
smaller than the corresponding values for ordinary osteoclasts,
further they were surrounded by numerous histiocytes. Therefore,
we believe that these multinucleate giant cells were histiocytic
in origin. We speculate that the histiocytes first phagocytose
serum bilirubin in the bone marrow and actively proliferate,
with some of them fusing with each other to form multinucleate
giant cells, causing bone resorption. This phenomenon suggests
that bilirubin may induce osteoprosis by activating histiocytes,
a mechanism similar to that of polyethylene- induced particle
disease causing loosening of the stem after artificial joint
replacement7), 8). The major cause of bone resorption
around total joint protheses is the inflammatory response to the
wear debris of polyethylene. Particles derived from the wear
debris cause macrophage activation and phagocytosis. Gallo et al7) stated that aseptic loosening and osteolysis after hip
arthroplasty is caused predominantly by osteoclasts, mediated
mainly by an osteoprotegerin ligand(RANKL) and TNF-α. RANKL has been shown to be
expressed in activated macrophages, osteoblasts, and lymphocytes7).
We believe that bilirubin may also behave similarly to these
particles.
This is presumably the first report of the
histological findings in the bone in patients with marked and
persistent hyperbilirubinemia.
Reference :
-
Dinesh MD, FRCA, Mohammed MD et al; The Alagilles
syndrome and its anaesthetic considerations: Paediatric
Anesthesia Vol 8(1) 79-82, 1998.
-
G. Maldini, E Torri, A Lucianetti et al; Orthotopic liver
transplantation for Alagille syndrome: Transplantation
Proceedings Vol 37(2) 1174-1176, 2005.
-
A. Bagur, C. Mautaken, J. Findor et al; Risk factor for
the development of vertebral and total skeleton osteoporosis in
patients with primary biliary cirrhosis: Calcif Tissue Int 63
385-390, 1998.
-
N. Guanabens, A. Pares, I Ros et al; Severity of
cholestasis and advanced histological stage but not menopausal
status are the major risk factors for osteoporosis in primary
biliary cirrhosis: J Hepatol Vol42(4) 573-577, 2005.
-
S. Ormarsottir, O Ljunggren, H Mallmin et al; Increased
rate of bone loss at the femoral neck in patients with chronic
liver disease: Europ J Gastroenterol & Hepatol Vol 14(1), 2002.
-
C. Janes, Dickson ER, Bond S; Role of hyperbilirubinemia
in the impairment of osteoblast proliferation associated with
cholestatic jaundice: J Clin Investigation Inc. Vol(95)
2581-2586, 1995.
-
Gallo J, Kaminek P, Ticha V et al; Particle disease. A
comprehensive theory of periprosthetic osteolysis: a review:
Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub.
146(2):21-28, 2002.
-
A V Parwani, B Yang, D P Clark et al; Particle disease:
cytopathologic findings of an unusual case: Diagn Cytopathol.
Vol31(4):259-262, 2004.
|