|
Abstract:
Backround. Hence understanding in vivo cell functions by means of osteoblast culture experiments, tissue engineering in orthopaedics has been developed. The aim of this study is to elicitate spreading characteristics of cultured human osteoblastic cells into 3 dimensional architecture of various graft materials.
Methods. Three different graft materials which are already in clinical use were investigated as an osteoblast carrier: 1- Allogenic human cancellous bone. 2-Coralline hydroxyappatite. 3- Biphasic tricalcium phosphate/hydroxyappatite. Human bone marrow was obtained from iliac crest. Thereafter mesenchymal stem cells cultured and expanded. Osteoblastic phenotype is determined by Alkalen phosphatase activity, Von Kossa stain and CBF 1 gene test.
Results. After primary culture, cells were seeded on grafts. In phase contrast microscopy, cells had attached and proliferated on all surfaces of grafts. On day 21 of secondary culture, scanning electron microscopy was performed. Electron microscopy revealed that osteoblasts mithotically expanded upon the surfaces of all grafts with underlying abundant collagen matrix. Cells were visible on the entire 3 dimensional architecture. Cell concentration on the surfaces is subjectively equal.
Conclusions. Our study demonstrated that, these graft materials were equally biocompatible and osteoconductive. Also composition of these grafts with osteoblasts, creates novel living ceramics. The composite is reliable and stable for in-vitro conditions and promises a new perspective for clinical applications of these three well known graft materials
J.Orthopaedics 2011;8(1)e6
Keywords:
Osteoblast; graft; primary culture
J.Orthopaedics 2011
Introduction:
Matrices serve as a scaffold for attachments, proliferation and new matrix synthesis to osteoblastic cells via its biocompatible surfaces. Desired functions of all matrix types are biocompatibility, degredability and considerable mechanical properties. Plenty more matrices and graft materials are currently used for lots of reasons in orthopaedic surgical applications such as filling bone defects, enhancement of fracture healing, spinal fusions.1
Allograft bone, biphasic tricalcium phosphate/hydroxyappatite (TCP/HA), coral derived implants and synthetic biodegredable matrices have majority in this applications. Each matrices has distinct 3 dimensional (D) architecture, porosity and interconnection properties which are closely related bone formation thoroughout entire graft volume.2,3 Bone marrow contains mesenchymal stem cells (MSC) that have capable to differantiate osteoblastic lineage. MSC’s could have been differantiated and extensively expanded in appropiate in vitro culture conditions prior implantation in vivo.3
In recent years many osteoconductive materials have been widely examined for ideal matrix and osteoblast composite both in vitro and in vivo.4,5,67,8 Morever allograft bone, coralline hydroxyappatite and biphasic TCP/HA are proven graft materials as bone substitutes for clinical applications.4 The in vitro ability as a carrier for osteoblasts of above mentioned graft types which are already in the market has not clearly defined yet and therefore the first aim of this experiment is to produce an composite graft which is osteoconductive with allograft, coralline hydroxyappatite, synthetic TCP/HA and osteogenic with human bone marrow derived osteolast cell culture. Also we want to elicitate spreading characteristics of osteoblastic cells to entire 3D architecture and in order to understand in vivo bone formation in scaffold and to compare these grafts as osteoblast carrier for in vivo applications.
Material and methods
Grafts and properties
Three different commerically available graft materials were used in present study (Figure1).
1-Allogenic human cancellous bone derived from human cancellous bone, contains hydroxyapatite and collagen, interconnection porosity (Tuttoplast, Tutogen Medical, GmbH Germany).
2-Coralline hydroxyappatite: Coral skeleton covered by hydroxyapatite, interconnection porosity (Interpore 500, Proosteon, Interpore International, USA).
3-Synthetic, biphasic TCP/HA: 60/40 ratio of hydroxyapatite/ß-tricalcium phosphate, interconnection porosity (Camceram, Cam Implants BV, Holland)(Figure 1).
Figure 1
Human bone marrow derived osteoblast culture
After permission of local ethic comitee, using a jamshidi needle, 4 ml human bone
marrow aspirated from two different sites of posterior iliac crest (each for 2 ml and patients mean age was 21) who has underwent posterior spinal insturmentation. Marrow placed in a 20 ml tube contains Dulbecco’s minimal essential medium (DMEM) primary media and centrifuged at 500g 10 min at + 4ºC for isolation of cells.
Cells were washed with phosphate buffered saline (PBS) and thereafter cells were plated on to two T 75 flasks. Cells were undistrubed for 5 days to provide cell attachment. In primary culture period cells were fed with primary medium contains K–MEM, peniciline 100U/ml, streptomycine 100Ng/ml and %10 fetal bovine serum, dexametasone, P Gliserophosphate, L Ascorbic acid. Medium was changed every three day and cells cultured in humidified atmosphere (% 95 air and % 5 CO 2 ) at 37 ºC. Cells were observed with a phase contrast microscope (Nikon, Japan) daily and spindle shaped cells were seen. Primary culture reached confluency about 14th day of initial seeding.
Flasks were washed two times with PBS adherent cells were treated with trypsin and Ethylenediaminetetraacetic acid (EDTA) and counted with coulter counter then prepared for secondary culture and loading. Cell loading on grafts after confluency was reached, cells were removed from the flasks with trypsinization and suspensed with 2 ml secondary medium. Coulter counter was used for assesing of plating density. Graft materials were placed in well plates and for each, cell suspension were added in medium with the density of 1.2X106 cell/cm2. Cells were fed every three day with complete medium contains K–MEM, peniciline, streptomycine and %10 fetal bovine serum, dexametasone, P Gliserophosphate, L- Ascorbic acid. Cell and matrix composite were evaluated daily with phase contrast microscopy. The composite prepared for histology and Scanning Electron Microscopy (SEM) observation at 21th day of secondary culture.
Characterisation of osteoblastic phenotype
Three tests were performed to detect the osteoblastic phenotype of the cultured cells; alkaline phosphatase (ALP) activity, quantification of bone nodules with von Kossa stain, CBF_1 gene expression, measurement of intracellular ALP was used for identification of osteoblastic cells.
Briefly cells were collected by a cell scraper in second week of secondary culture. After three times rinsing with PBS, centrifugation was performed. Cells were lysed and aliquot was used for spectroscopic determination of total protein and ALP. Para nitrophenol was used as a substrate and at the same time ALP activity was measured in fibroblast culture as a control.9
Bone nodule formation
In the secondary culture period, cells were maintained for 21 days for bone nodule formation. On the last three days of culture, 5 milimeter (mm) inorganic phosthate added in the medium.9 On day 21 in situ von Kossa stain was performed for visualization of bone nodules.
CBF_1 gene expression is carried out using Reverse transcription polymerase chain reaction (RT-PCR) method. Briefly cell lines at the secondary culture were used for DNA isolation. RT-PCR amplification with specific primer was confirmed the CBF_1 gene.
Scanning Electron Microscopy
After 21 days of secondary culture, graft+cell composites were prepared for SEM. For each graft material, eight samples were analyzed. At least ten photos for each sample were collected for documentation and evaluated with two independent observer. Graft materials were washed with PBS for three times and fixed in gluteraldehyte in 0.1M cocodylate buffer pH(7.4) and left overnight in it. Samples were dehydrated in graded alcohol 100/70/50/25%).
After sputter coating with a thin (250 angstrom) layer of gold (Hummer VII, Anatech Ltd. Alexandria, VA, USA). Samples were examined in a SEM (JSM-6400, JEOL, JAPAN) with 20 kilowatt accelerating voltage.
Results:
Bone marrow derived cells were showed osteoblastic phenotype in the early stages of secondary culture and maintained during the experiment period. Classical spindle shaped cells were confluent on tissue plastic in the primary culture period. ALP activity of cultured cells is evidently high when compared with fibroblast culture on day 21,27/1.7 microunit/miligram (mu/mg) protein respectively. Osteoblast like cells were showed bone nodule formation on the tissue culture plastic with von Kossa stain. CBF_1 was a positive indicator of osteoblastic phenotype and determined at the secondary culture. As a result phenotypic stability of plated cells was proved as well.
Phase contrast microscopy
After seeding on the graft materials, cells were gradually proliferated both on tissue plastic and graft material. Cells on the flask surface accumulated around the edges of the grafts that suggesting the affinity of these cells to the biocompatible surface properties of the grafts. At day seven, cells were clearly evident on the surface and in the porous structure. Cells were mitotically expanded on the surfaces during the incubation period. As culture time progressed, cell spreading and surface covering gradually increased. Cells were multilayer on the surface of the grafts and produced extracellular matrices in some part of graft surfaces (Figure 2).
at day 21, cells were found to be nearly confluent on the surfaces, multilayer sheets and extracellular matrix formation are also seen. At day 21, cells were found to be nearly confluent on the surfaces, multilayer sheets and extracellular matrix formation are also seen.

Figure 2
Scanning electron microscopy
Semiquantitative evaluation scale was used for evaluating of cell-material relationship. SEM photographs were examined for six paramaters; 1-cell layer, 2-confluency, 3-extracellular matrix synthesis, 4-cell distribution, 5-cell morphology, 6-cellular interactions. Achieved points of each graft material were determined (Table). Spindle and round shaped cells onto surface and pores with various concentrations were observed. Most of the cells have spindle morphology, round cells were not dominantly seen. Cells were flattened, firmly attached to the graft and these were visible on the entire 3D architecture. This data suggested that osteoblasts mitotically expanded throughout of all grafts with underlying collagen matrix. Dense extracellular matrix formed up on surface and nearly covered the entire graft volume. Drying procedure caused rupture of matrix in some areas.
Cells attached onto surface and each other with filopodal processes and extracellular matrices. Cells were connected with adjacent cells with bipolar or filopodial processes. Plated cells were determined with subconfluent densities and therefore concentration on the surfaces of each material is subjectively equal. Cell loaded coralline hydroxyappatite, biphasic TCP/HA have similar electron microscopic appearances with allogenic bone. But cells on the surface of allogenic bone showed higher rate of multilayer cell lines and dense cell packages and accumulations which suggested nodule formation (Figure3 a-b-c). On the surface of grafts, cells appeared to be flattened and oriented in a parallel way, and arranged as straight flat sheets whereas in pores cell spreading seems to be irregular.
TABLE
|
Cell layer |
Confluency |
ECM |
Cell distribution |
Cell morphology |
Cellular interactions |
Allograft |
2 |
3 |
3 |
3 |
3 |
2 |
Coralline HA |
1 |
3 |
3 |
3 |
2 |
2 |
Biphasic TCP/HA |
1 |
3 |
3 |
3 |
2 |
2 |
Semiquantitative evaluation scale for cell-material relationship with SEM.
Cell layer: enitrely multilayer (3), partially multilayer(2), monolayer (1).
Confluency: confluent (4), subconfluent (3), semiconfluent (2), sparse (1).
Extracellular matrix synthesis: abundant (3), moderate(2), insufficent (1).
Cell distribution: equal at all sites include pores and surface(3), dominantly on surface (2), rare even on surface (1).
Cell morphology: dominantly spindle - flattened (3), equally spindle and rounded (2), dominantly rounded (1)
Cellular interactions: firm bonding with other cells (3), filopodal connections (2), seperate low cellular interactions (1).
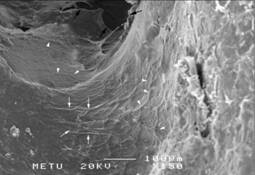 
Figure 3a Figure 3b

Figure 3c
Discussion
Allograft bone, porous hydroxyappatite and biphasic TCP/HA have been extensively researched in human clinical applications like traumatic bone defects, tumour resections, spinal fusions, hip and knee surgery.1 As the unique bone graft that contains osteoblasts and its precursors, autogenic cancellous bone is the gold standard for grafting procedures. The ultimate objective of growing cultured human osteoblasts onto grafts is to enhance their potential as bone graft substitutes. 3,10 In vivo experiments of cell loaded matrices
revealed higher rate of union, bone healing response and forming de novo bone.11,12
Human bone marrow derived osteoblast are capable to attach, growth, proliferate upon these graft materials. Our results steated that cultured cells were sufficiently repopulated in the entire volume of the investigated matrices. Osteoblasts spread of the entire 3D architecture of grafts with subconfluent densities. Consequently formation of osteogenic and osteoconductive composite graft was achieved with these commerical graft materials. Advantages of these composites are; commercially available matrices were used, no need to specially manufactured grafts, suitable for human clinical trials because grafts are already in use, proven for grafting procedures and provides mechanical stregth.
Disadvantages of these composites are; difficult to interprete exact number of loaded osteoblasts on grafts, when compared with some other carriers like demineralised bone, repopulated cells were in low density, for allogenic bone disease transmission risk is present. Marrow derived osteoblastic cells easily attached to demineralized bone, proliferated markedly and reached confluency on above all surfaces of demineralized bone.13 Also multilayering of osteoblasts on demineralized spongious animal bone was reported.
Seemingly demineralized bone showed better performance of confluency and cell layering for in vitro conditions than conventional osteoconductive grafts.4,13 But demineralized bone has no mechanical strength. Specially prefabricated hydroxyappatite and calcium phosphate based constructs were also invesitgated for osteoblast transplantation.6,14,15,16,17,18 Overall data of these studies indicated that normal bone tissue was regenerated after grafting of the cell loaded osteoconductive grafts. Some other novel resorbable graft materials like poly (L-lactic acid) and poly-(D,L-lactide) were investigated for osteoblast transplantation, but routine clinical applications in orthopaedic practice were not established yet.5,8 As a result; tissue engineering approaches of cultured marrow cells with these matrices may provide an alternative method of cell transplantation for patients.18
Ducheyne stated that pluripotential cells have intrinsic capability to differantiate along the osteoblastic pathway, but osteogenic potential of these cells is also stimulated by the bioactive material surface.7,19 Also noted that porous structure is essential to stimulate differantiation of precursors to osteoblasts. In vitro cell adhesion is mainly under control of graft topography and antigenity.4,19,20 For in vivo conditions, presence of the HA surface supports osteoblastic differentiation of marrow stromal stem cells, afterwards the cells had initiated partially mineralized osteoid formation directly onto the surface. 16,17 In our study; similar porous structure and hydroxyappatite surface features of three grafts led to enhanced cell attachment and therefore almost the effectively same composite artificial grafts could have been produced. Surface of all matrices were HA coated therefore equal SEM results are mainly related to this factor.
Present study suggest that allograft bone showed better performance for multilayering and repopulated cell number. This feature is possibly related by extracellular matrix (collagen) component of allograft bone. Because collagen facilitates deposition of hydroxyappatite and provides additional sites for cell attachment.2 Therefore presence of hydroxyappatite and collagen may potantialize the effectiveness of allograft bone as a carrier. We believed these composites are reliable and stable for in vitro conditions and promises new clinical application for these three well known graft materials as a “novel living grafts”. Graft materials herein served for carrying osteoblast equally via its biocompatible and osteoconductive properties. These compositest may be effectively similar to autogenous bone. But further investigations are required to determine its in vivo bone healing effect.
References
1. Cornell CN. Osteoconductive materials and their role as substitutes for autogenous bone grafts. Orthop Clin North Am. 1999;30(4):591-598.
2.Fleming JE, Cornell CN, Muschler GF. Bone cells and matrices in orthopaedic tissue engineering. Orthop Clin North Am 2000; 31(3):357-374.
3. ROC, Triffitt JT. Future potentials for using osteogenic stem cells and biomaterials in orthopedics. Bone 1999; 25(2) supp;5-9.
4. Begley CT, Doherty MJ, Hankey DP, Wilson DJ. The culture of human osteoblasts upon bone graft subsitues. Bone 1993;14: 661-666.
5. Cai K, Yao K, Lin S, Yang Z, Li X, Xie H, Qing T, Gao L. Poly (D,L-lactic acid) surfaces modified by silk fibroin: effects on the culture of osteoblast in vitro.
Biomaterials 2002;23:1153-1160.
6. Dong J, Kojima H, Uemura T, Kikuchi M, Tateishi T, Tanaka J. In vivo evaluation of novel porous hydroxyappatite to sustain osteogenesis of transplanted bone marrow derived osteoblastic cells. J Biomed Mater Res 2001;57:208-216.7. Ducheyne P, Qui Q. Bioactive ceramics: the effect of surface reactivity on bone formation and bone cell function. Biomaterials 1999; 20:2287-2303.
|







